How many unpaired electrons are present in Mn^(2+) ion ? How do they influence magnetic behaviour of Mn^(2+) ion?

Magnetic moment of `Cr^(+2)(Z=24),Mn^(+2)(Z=25)` and `Fe^(2+) (Z=26)` are x,y,z. they are in order - YouTube
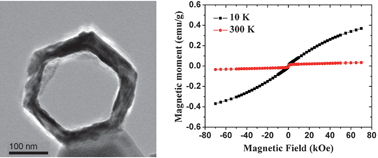
Single-crystal like mesoporous ZnO:Mn2+ nanorings of high optoelectronic quality formed by self-assembly of nanoparticles in an ultrasonic hydrolysis process - Nanoscale (RSC Publishing)

In the fallowing reaction: Cr2O7^2 - (aq) + SO3^2 - (aq) + 8H^+→ 2Cr^3 + + SO4^2 - + H2O the stoichiometric coefficient of SO3^2 - is:

calculate the number of unpaired electrons in Ti3+ , Mn2+ and calculate the spin only magnetic - Brainly.in
12. Exerimental value of magnetic moment of Mn2+ complex is 5.96BM.This indicates a)axial and orbital motion of electron in same direction b)axial and orbital motion of electron in opposite direction c)electron does

Among V(Z = 23) , Cr(Z = 24) , Mn(Z = 25) and Fe(Z = 26) , which will have the highest magnetic moment?




![Calculate the magnetic moment of Mn^(2+) ion. [Atomic number of Mn=25] Calculate the magnetic moment of Mn^(2+) ion. [Atomic number of Mn=25]](https://doubtnut-static.s.llnwi.net/static/web-thumb/643905480_web.png)